AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
Back to Blog
Salt bridge battery cathode and anode7/27/2023 So thinking about an ox reminds us that oxidation occurs at the anode and thinking about a red cat reminds us that reduction Reduction occurs at the cathode is to think about twoĪnimals, an ox and a cat. 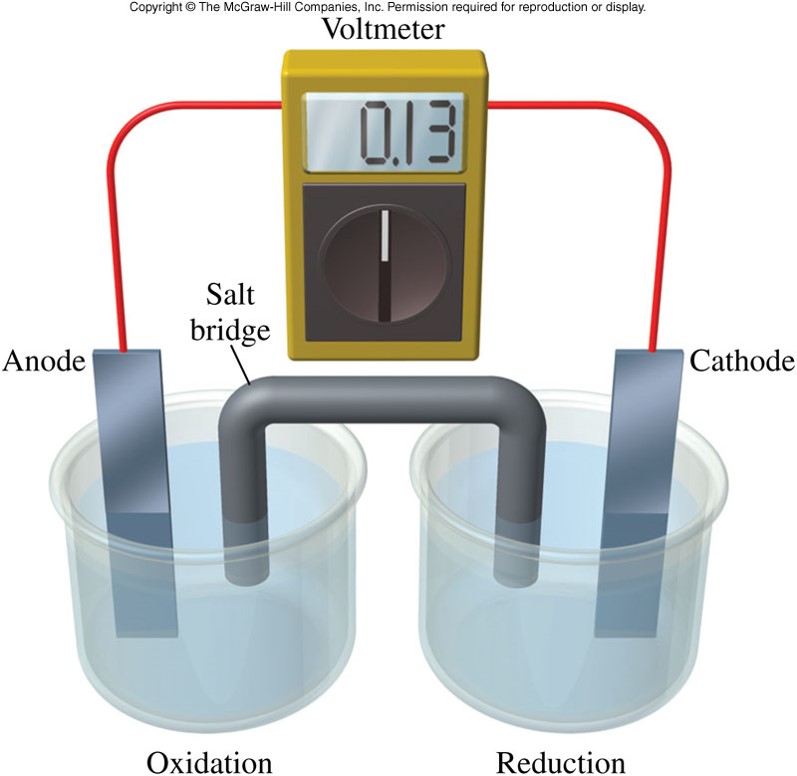
A good way to remember that oxidation occurs at the anode and And the concentration of copper 2+ ions in solution will decrease. So as copper 2+ cations gained two electrons to turn into solid copper, over time, the amount of copper, solid copper electrode will increase. And the amount of zinc 2+ cations in solution will increase. The amount of the solid zinc electrode will decrease. So solid zinc turns into zinc 2+ cations. The anode is where oxidation takes place. So we can think about what's gonna happen to our cell over time. I went ahead and rewroteĮach half reaction to remind us of what's going The electrode where reduction takes place is called the cathode. At the surface of this copper electrode, the copper 2+ ions come in contact with two electrons and are 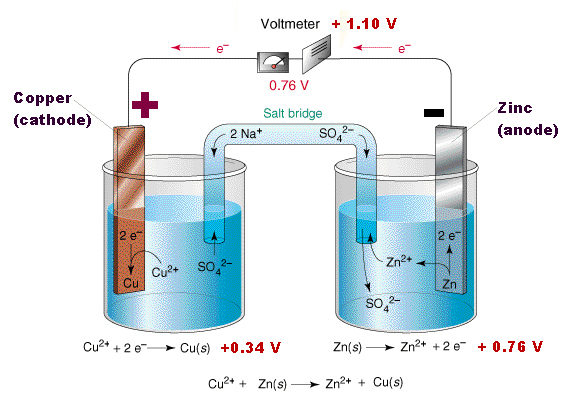 
We know that electronsĪre moving in this wire that connects the two compartments. There are copper 2+ ions and sulfate anions in aqueous solution. Next, let's look at theĬompartment on the right, which is a beaker that contains a one molar solution of copper sulfate. Oxidation takes place is called the anode. So imagine we have movement of electrons going from the compartment on the left towards the compartment on the right. So those electrons will move in this wire that connects When zinc turns into zinc 2+, two electrons are lost. We saw from the oxidation half reaction that solid zinc is oxidized and turns into zinc 2+ cations. Therefore, there are zinc 2+ ions and sulfate anions in aqueous The compartment on the left, which is a beaker that contains a one molar aqueous Galvanic or voltaic cell, let's look at a diagram of this cell and see how things actually work. Over the half reactions in detail for this zinc-copper And by electric current, we're talking the flow of electrons. Therefore, a thermodynamically favorable redox reaction is used to generate an electric current in the wire. And the two compartmentsĪre connected with a wire. However, in a galvanic or voltaic cell, each half reaction gets So that's what would happen if we did this reaction in only a single compartment. Put a piece of solid zinc in an aqueous solution of copper 2+ ions, we would see copper metal form on the zinc metal and zinc 2+ ions would form in solution. Delta G naught for this reaction at 25 degrees Celsius or room temperature is less than zero, which means this reaction is thermodynamically favorable. In this redox reaction, zinc metal is oxidized to zinc 2+ cations and copper 2+ cations are Goes to zinc 2+ cations and solid copper. That gives us a solid zinc plus copper 2+ ions Half reactions together, we have two electrons on the reactant side and two electrons on the product side. 
So loss of electrons is oxidation and gain of electrons is reduction. Which half reaction is which is to think about LEO the lion goes GER. Since there's a decrease or a reduction in the oxidation numberįrom plus two to zero, this is the reduction half reaction. So the copper 2+ cation has an oxidation number of plus two and solid copper has an oxidation number of zero. That this is the reduction half reaction is to lookĪt the oxidation numbers. In this half reaction, the copper 2+ cation gains two electrons to turn into solid copper. And here is the other half reaction that we're going to see in As a quick review, the other way to tell that this is the oxidation half reaction is we go from an oxidation number for solid zinc of zero to an oxidation numberįor zinc 2+ of plus two and increase in the oxidation In the first half reaction, zinc metal loses two electrons to turn into the zinc 2+ cation. Before we look at a diagram of a galvanic or voltaic cell, let's first look at the half reactions that are going to be used in the cell. Galvanic cells, which are also called voltaic cells, use a thermodynamically favorable reaction to generate an electric current.
0 Comments
Read More
Leave a Reply. |
 RSS Feed
RSS Feed
